Li-In alloy is one of the most popular Li alloys used in sulfide-SSE batteries ( 9 – 11). In addition, the safety of batteries is greatly improved because of the suppression of lithium dendrite growth. The degradation of SSEs caused by active Li can be greatly relieved. This substitution can result in two effects. Li alloys with a suitable operating potential are applicable substitutes for Li as anodes in solid-state batteries. However, the complicated pretreatment process will largely increase the difficulty of preparation and raise the cost. The buffer layer can also be the artificial solid electrolyte interface (SEI) formed by the pretreatment of Li or the SSE ( 7, 8). Meanwhile, the internal resistance of batteries will increase with the introduction of more interfaces. A buffer layer, such as Li 3PS 4 ( 5) or 0.75Li 2S-0.24P 2S 5-0.1P 2O 5 ( 6), can effectively enhance the stability between the electrolyte and highly active Li anode. Two strategies have been proposed to solve the instability problem of electrolyte with Li: the protection of Li by a buffer layer and the substitution of Li by another anode with better compatibility to sulfide electrolytes. Nevertheless, sulfide electrolytes suffer from their incompatibility with Li. Sulfide SSEs, such as Li 10GeP 2S 12 (LGPS) and Li 6PS 5Cl, with high ionic conductivities and moderate Young’s modulus can result in high rechargeability and low internal resistance of batteries ( 3, 4). SSEs act as a Li-ion pathway within the battery and a separator between the cathode and anode. This work provides an applicable anode selection for all-solid-state LSBs and promotes their practical procedure. 
Furthermore, a Li-S full cell with a low negative-to-positive ratio of 1.125 delivered a specific energy of 541 Wh kg −1. An all-solid-state LSB comprising Li 0.8Al alloy anode and melting-coated S composite cathode functioned steadily for over 200 cycles with a capacity retention of 93.29%. The Li 0.8Al alloy anode showed excellent compatibility toward the Li 10GeP 2S 12 (LGPS) electrolyte, as verified by the steady Li 0.8Al-LGPS-Li 0.8Al cell operation for over 2500 hours at 0.5 mA cm −2. 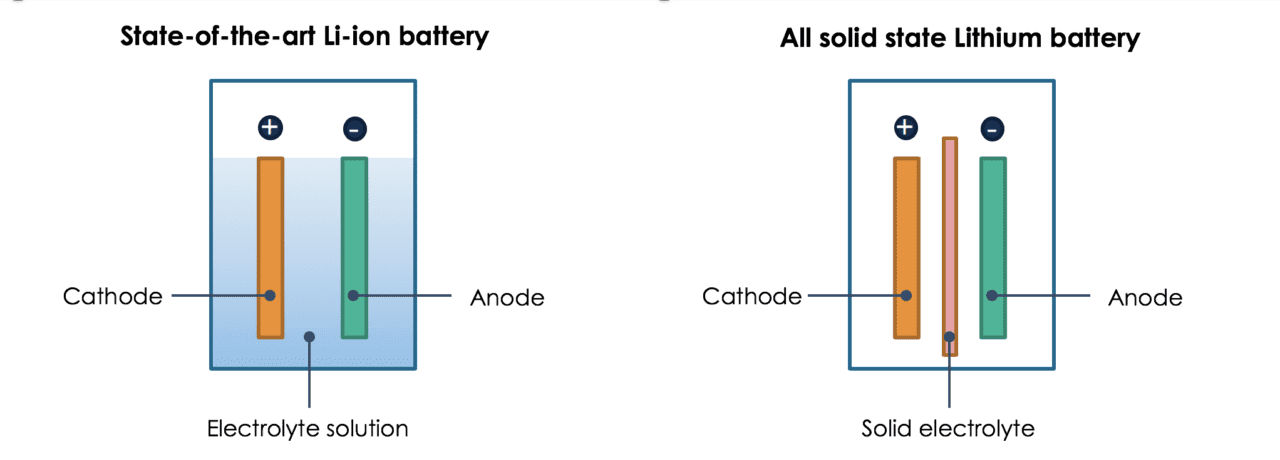
A biphasic lithiation reaction of Al with modest volume change was revealed by in situ characterization. Aluminum with appropriate potential, high-capacity, and electronic conductivity can alloy with Li spontaneously and is proposed herein as a carbon-free and binder-free anode of an all-solid-state Li-S battery (LSB). Incompatibility of electrolytes with Li anode impedes the application of solid-state batteries.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
